- Blog
- Nvidia 3d tv unable to contact licensing server
- Revit 2011 license error
- Romhustler esa protected rom
- Extreme ghostbusters gba
- Will amiga os 3-9 work with winuae
- Tech tool pro 9
- Vintage bodybuilding books for sale
- Mastercam cost
- Hawaii music festival 2021
- Bluestacks 2 app player download
- Is pete droge still performing
- Electronics recycle center near me
- Jcb 3cx buckets for sale
- How to setup usb bitcoin miner
- Oxygen in zno xps peak
- Pes 2018
- Tales from the yawning portal zombie
- The jonbenet bartleby music
- The office season 8 episode 17
- Native instruments service center update
- Free symantec endpoint protection 14 download full version
- Koihime musou doki otome darake no sangokushi engi combat
- Corel motion studio 3d keygen generator download
- Python file extension
- Activ energy battery charger sold green light
- Il corsaro nero vasco rossi
- Magix music maker 2021
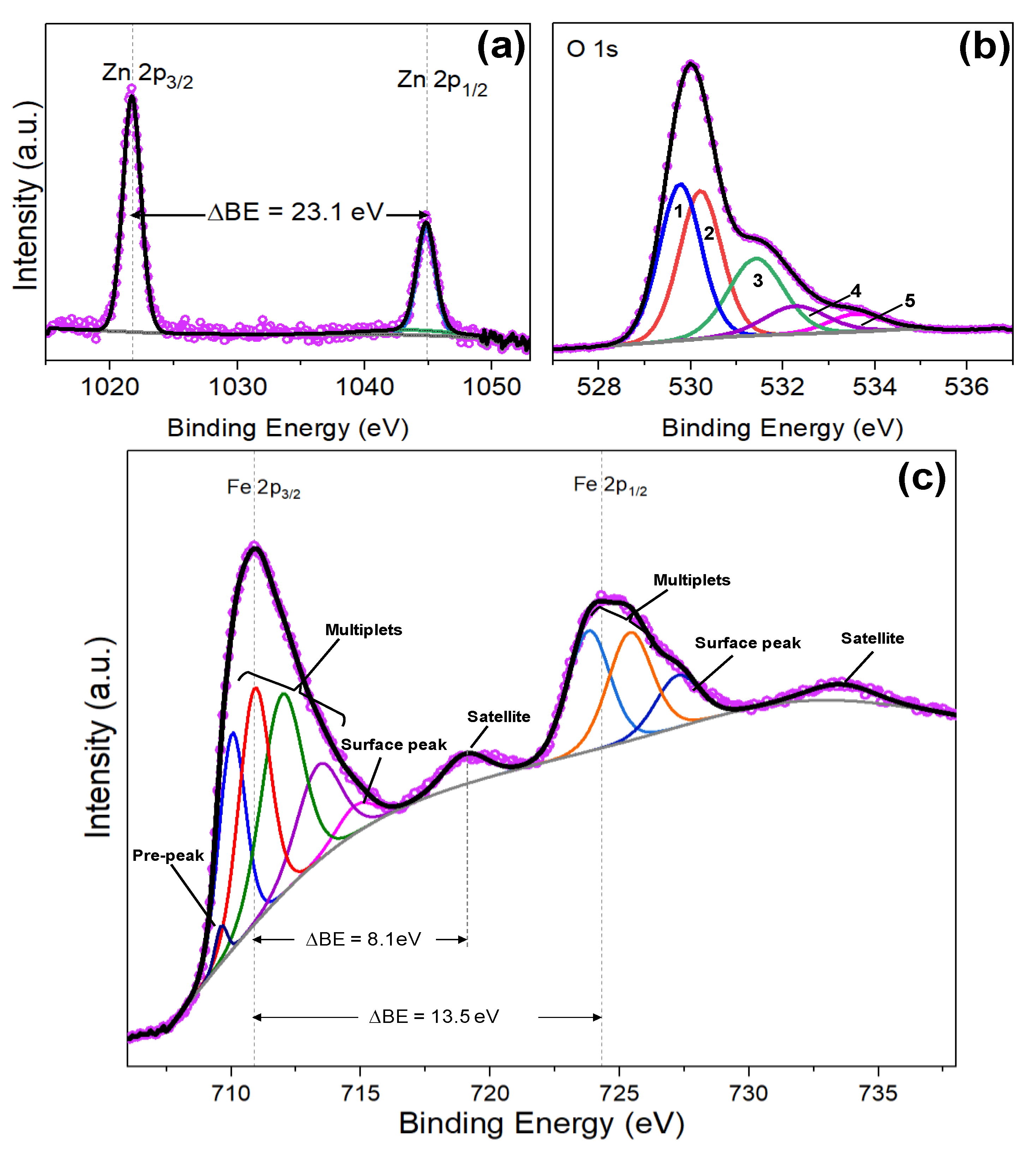
Oxygen is colorless, odorless, and tasteless in its gaseous form, and condenses to pale blue liquid and solid forms. Accounting for one-fifth of the earth’s atmosphere, oxygen combines with most elements and is a component of thousands of organic compounds. Zn 2p3/2 XPS spectra of the as-prepared ZnO nanoparticles (a) ZnO. Oxygen is critical for life on Earth, produced by plants during photosynthesis and necessary for aerobic respiration in animals. The results indicated that the defect sites (Zn interstitials, oxygen vacancy. Oxygen, the "elixir of life", was discovered by Joseph Priestly and Carl Wilhem Scheele independently of each other in the 1770’s.
Oxygen in zno xps peak tv#
Zinc oxide is used in plastics, cosmetics, and wallpaper, while zinc sulfide is employed in luminescent dials, TV screens, and fluorescent lights.Discoverer: Joseph Priestley/Carl Scheele It is also used to form numerous metal alloys such as brass, bronze, and aluminum solder. Zinc’s principle use is to galvanize iron and steel, preventing corrosion. It is an essential element in the growth of plants and animals, and altered biological concentrations of zinc can lead to severe health problems. This bluish-white metal is brittle at ambient temperatures, but becomes malleable between 110–150☌. Marggraf is credited for the first isolation of zinc from the charcoal reduction of calamine in 1746. So the range of XPS survey scan was kept from 0-1100 eV in order to find the surface composition of upper layer of ZnO/Cu/ZnO XPS. Zinc alloys have been used for centuries although, A. Most elements occurring in the nature have photoelectron peak from 0 to 1100 eV. Use Wagner plot to help assign chemistry.Use X-ray induced Zn LMM Auger peaks, which have a larger shift with chemical state.Similarly, Zn3p (very high kinetic energy) may be observed even if Zn2p is not.The higher energy peak at 531.9 eV has been observed earlier 1416 and has been interpreted as defect oxygen or the oxygen containing surface contaminants. If zinc is buried (under carbon, for example), Zn Auger peak may be observed even if Zn2p is not (due to difference in electron kinetic energy). The O1s peak at 529.9 eV contributed to the O ions in the ZnO lattice and is surrounded by Zn ions and hence indicates the bonding of ZnO.Zn2p peaks often accompanied by Auger peak at Kinetic Energy ~990eV.Peak width may broaden in the presence of more than one Zn species.the only difference i get is that the O1s spectrum of only ZnO NPs give a sharp peak 530eV with rather. Zinc shows only a small binding energy shift in the Zn2p 3/2 region (1021eV - 1023eV, modal value). I want to confirm the presence of Zn(OH)2 in those samples by XPS.ZnO has an unusual O1s spectrum with two peaks, possibly indicating either Zn(OH) 2, ZnCO 3,.Zn2p peak has significantly split spin-orbit components (Δ metal=23eV). Figure 8c shows the high resolution core level O1s XPS spec- trum of the representative ZCNT0.1 NCs and hence it shows the strong peak at 530.47 eV assigned due to the characteristic binding energies for lattice oxygen of ZnO and again it deconvoluted into two peaks at 532.07 eV and 533.13 eV corresponding to chemisorbed water and COH.There are bigger chemical shifts observed for Zn LMM compared to Zn2p.Collect principal Zn LMM peak as well as Zn2p.Chemical state differentiation can be difficult with XPS only.There are small binding energy shifts of some compounds compared to zinc metal.
- Blog
- Nvidia 3d tv unable to contact licensing server
- Revit 2011 license error
- Romhustler esa protected rom
- Extreme ghostbusters gba
- Will amiga os 3-9 work with winuae
- Tech tool pro 9
- Vintage bodybuilding books for sale
- Mastercam cost
- Hawaii music festival 2021
- Bluestacks 2 app player download
- Is pete droge still performing
- Electronics recycle center near me
- Jcb 3cx buckets for sale
- How to setup usb bitcoin miner
- Oxygen in zno xps peak
- Pes 2018
- Tales from the yawning portal zombie
- The jonbenet bartleby music
- The office season 8 episode 17
- Native instruments service center update
- Free symantec endpoint protection 14 download full version
- Koihime musou doki otome darake no sangokushi engi combat
- Corel motion studio 3d keygen generator download
- Python file extension
- Activ energy battery charger sold green light
- Il corsaro nero vasco rossi
- Magix music maker 2021
